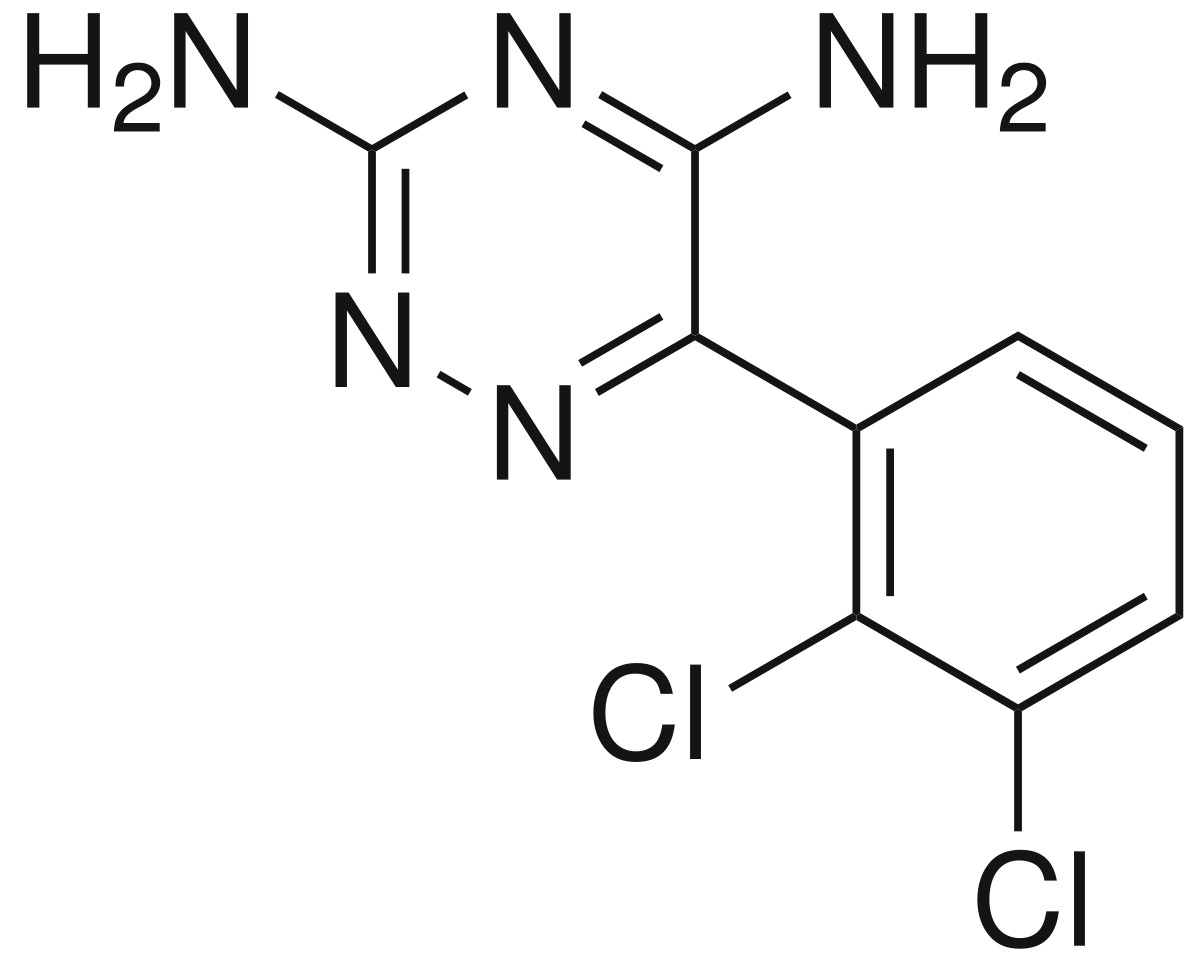
Stevens‐Johnson syndrome and toxic epidermal necrolysis with antiepileptic drugs: An analysis of the US Food and Drug Administration Adverse Event Reporting System - Borrelli - 2018 - Epilepsia - Wiley Online Library

Teva Announces Launch of a Generic Version of NuvaRing® (etonogestrel and ethinyl estradiol) Vaginal Ring in the United States | Business Wire

Stevens‐Johnson syndrome and toxic epidermal necrolysis with antiepileptic drugs: An analysis of the US Food and Drug Administration Adverse Event Reporting System - Borrelli - 2018 - Epilepsia - Wiley Online Library

Bioequivalence Between Generic and Branded Lamotrigine in People With Epilepsy: The EQUIGEN Randomized Clinical Trial. - Abstract - Europe PMC

Bioequivalence Between Generic and Branded Lamotrigine in People With Epilepsy: The EQUIGEN Randomized Clinical Trial. - Abstract - Europe PMC